Make-Up Work

Look for the date(s) you have missed in class. Any handouts or assignments given will be posted below. It is your responsibility to find and complete all missing assignments.

Click HERE for December 2012 Make-Up Work/Assignments
January 1, 2013 to January 3, 2013
Electron Configuration Notes
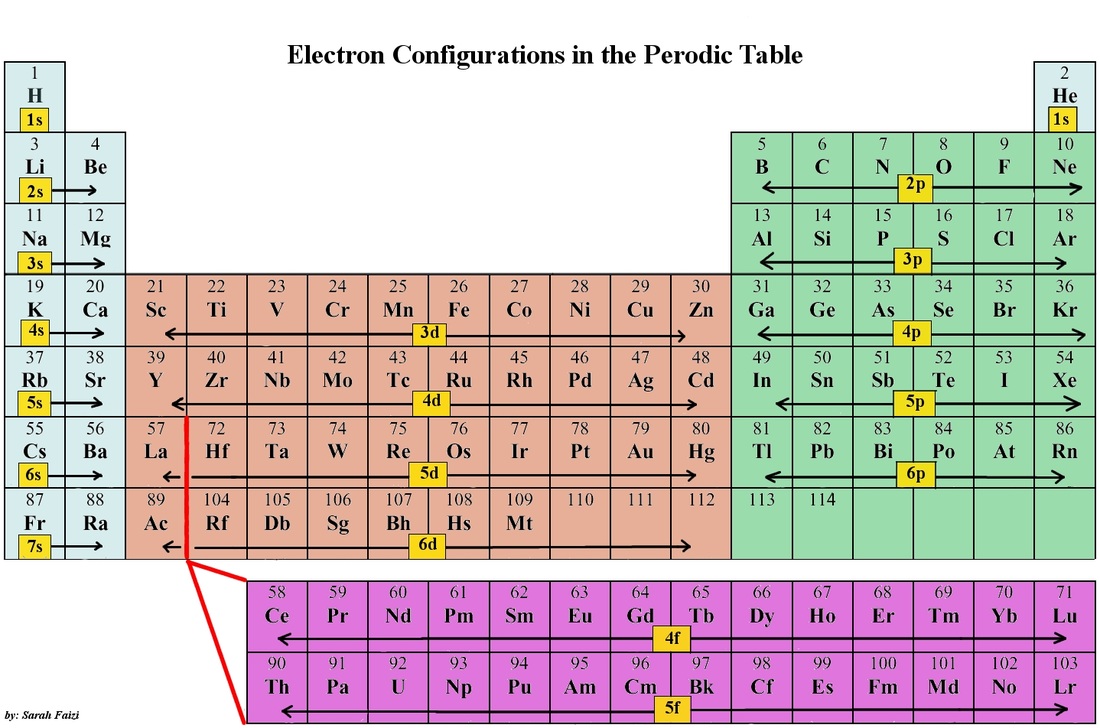
Electron Configuration: The way electrons are arranged around the nucleus.
The electron configuration of an atom is the representation of the arrangement of electrons that are distributed among the orbital shells and subshells.
Commonly, the electron configuration is used to describe the orbitals of an atom in its ground state, but it can also be used to represent an atom that has ionized into a cation or anion by compensating with the loss of or gain of electrons in their subsequent orbitals.
Many of the physical and chemical properties of elements can be correlated to their unique electron configurations. The electrons on the outer most shell, become the determining factor for the unique chemistry of the element.
Every element on the PT consists of an atom which is composed of protons, neutrons, and electrons.
Electrons exhibit a negative charge and are found around the nucleus of the atom. Electron orbitals are the position of the electrons around the nucleus and is determined as the volume of space in which the electron can be found within 95% probability.
The four different types of orbitals s,p,d, and f have different shapes.
As stated, the electron configuration of each element is unique to its position on the periodic table. The energy level is determined by the period and amount of electrons by the atomic number of the element.
Orbitals on different energy levels are similar to each other, but they occupy different areas in space. For example, the 1s orbital and 2s orbital both have the characteristics of an s orbital (radial nodes, spherical volume probabilities, can only hold two electrons, etc.) but as they are found in different energy levels they occupy different spaces around the nucleus. Each orbital can be represented by specific blocks on the periodic table.
Review of Bohr Model
http://www.youtube.com/watch?v=STCrjxxRf-g
Quantum Mechanical Model
http://www.youtube.com/watch?v=cKzh5yeQGjA
Quantum Mechanical Model
1920’s
Werner Heisenberg (Uncertainty Principle) Louis de Broglie (electron has wave properties)
Erwin Schrodinger (mathematical equations using probability, quantum numbers)
Heisenberg uncertainty principle
It is impossible to determine simultaneously (at the same time) both the position and velocity (speed) of an electron or any other particle with any great degree of accuracy or certainty.
Three rules are used to build the electron configuration: Aufbau Principle (Electrons occupy orbitals of lower energy first.)
Pauli Exclusion Principle (An orbital can hold only two electrons and they must have opposite spin.)
Hund’s Rule (In a set of orbitals, the electrons will fill the orbitals in a way that would give the maximum number of parallel spins (maximum number of unpaired electrons)
Analogy: Students could fill each seat of a school bus, one person at a time, before doubling up.
Sites to Visit to Help Better Understand Electron Configuration:
https://www.khanacademy.org/science/chemistry/orbitals-and-electrons/v/electron-configurations
http://library.thinkquest.org/10429/low/eleconfig/electron.htm
http://electronconfiguration.info/
The electron configuration of an atom is the representation of the arrangement of electrons that are distributed among the orbital shells and subshells.
Commonly, the electron configuration is used to describe the orbitals of an atom in its ground state, but it can also be used to represent an atom that has ionized into a cation or anion by compensating with the loss of or gain of electrons in their subsequent orbitals.
Many of the physical and chemical properties of elements can be correlated to their unique electron configurations. The electrons on the outer most shell, become the determining factor for the unique chemistry of the element.
Every element on the PT consists of an atom which is composed of protons, neutrons, and electrons.
Electrons exhibit a negative charge and are found around the nucleus of the atom. Electron orbitals are the position of the electrons around the nucleus and is determined as the volume of space in which the electron can be found within 95% probability.
The four different types of orbitals s,p,d, and f have different shapes.
As stated, the electron configuration of each element is unique to its position on the periodic table. The energy level is determined by the period and amount of electrons by the atomic number of the element.
Orbitals on different energy levels are similar to each other, but they occupy different areas in space. For example, the 1s orbital and 2s orbital both have the characteristics of an s orbital (radial nodes, spherical volume probabilities, can only hold two electrons, etc.) but as they are found in different energy levels they occupy different spaces around the nucleus. Each orbital can be represented by specific blocks on the periodic table.
Review of Bohr Model
http://www.youtube.com/watch?v=STCrjxxRf-g
Quantum Mechanical Model
http://www.youtube.com/watch?v=cKzh5yeQGjA
Quantum Mechanical Model
1920’s
Werner Heisenberg (Uncertainty Principle) Louis de Broglie (electron has wave properties)
Erwin Schrodinger (mathematical equations using probability, quantum numbers)
Heisenberg uncertainty principle
It is impossible to determine simultaneously (at the same time) both the position and velocity (speed) of an electron or any other particle with any great degree of accuracy or certainty.
Three rules are used to build the electron configuration: Aufbau Principle (Electrons occupy orbitals of lower energy first.)
Pauli Exclusion Principle (An orbital can hold only two electrons and they must have opposite spin.)
Hund’s Rule (In a set of orbitals, the electrons will fill the orbitals in a way that would give the maximum number of parallel spins (maximum number of unpaired electrons)
Analogy: Students could fill each seat of a school bus, one person at a time, before doubling up.
Sites to Visit to Help Better Understand Electron Configuration:
https://www.khanacademy.org/science/chemistry/orbitals-and-electrons/v/electron-configurations
http://library.thinkquest.org/10429/low/eleconfig/electron.htm
http://electronconfiguration.info/